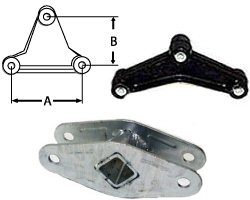
In electrochemistry, the anode is where oxidation occurs and is the positive polarity contact in an electrolytic cell. This seems appropriate because the anode is the origin of electrons and where the electrons flow is the cathode. What is the charge of an anode and cathode? The anode is regarded as negative in a galvanic (voltaic) cell and the cathode is deemed positive. This outward current is carried internally by positive ions moving from the electrolyte to the positive cathode (chemical energy is responsible for this “uphill” motion). Why cathode is positive in battery?Ī battery or galvanic cell in use has a cathode that is the positive terminal since that is where conventional current flows out of the device. Solution : During charging of lead accumulator, reverse reaction takes place. Which reaction occurs at the cathode of a lead storage battery?ĭuring charging of a lead storage battery, the following reaction occurs at the cathode. This action occurs since the charging current is usually greater than the current necessary to reduce the remaining amount of lead sulfate on the plates. How do lead acid batteries work?Īs a lead-acid battery charge nears completion, hydrogen (H 2) gas is liberated at the negative plate, and oxygen (O 2) gas is liberated at the positive plate. Depending on the state of charge (SoC), the cell may temporarily be lower after discharge than the applied voltage. To charge a sealed lead acid battery, a DC voltage between 2.30 volts per cell (float) and 2.45 volts per cell (fast) is applied to the terminals of the battery. What is the principle of lead acid battery?Īs sulphuric acid is used as an electrolyte in the battery, when it gets dissolved, the molecules in it are dispersed as SO 4 – (negative ions) and 2H+ (positive ions) and these will have free movement. Lead turns into lead sulfate at the negative electrode, electrons driven from positive plate to negative plate. Lead oxide (PbO 2), electrons added to positive plate Is anode positive or negative in lead acid battery? Why anode is negative in battery?Īnode is negative in an electrochemical cell because it has a negative potential with respect to the solution while anode is positive in an electrolytic cell because it is connected to the positive terminal of the battery. At the same time, sulfate from the acid is coating the plates and reducing the surface area over which the chemical reaction can take place. What is reduced in a lead-acid battery?īasically, when a battery is being discharged, the sulfuric acid in the electrolyte is being depleted so that the electrolyte more closely resembles water. For instance during discharging process, the cathode will react with the sulfuric acid and will give the electrolyte electrons i.e. What is oxidized in a lead-acid battery?ĭuring charging or discharging a lead acid battery both the positive and negative electrodes will undergo reduction and oxidation the same time. Discharging a lead-acid battery is a spontaneous redox reaction. When a lead-acid cell is producing electricity (discharging) it is converting chemical energy into electrical energy. A lead-acid battery is made up of a number of lead-acid galvanic (voltaic) cells connected up in series. As the battery charges, the sulfuric acid reacts with the lead in the anode and cathode to produce lead sulfate. The two electrodes are separated by an electrolyte of sulfuric acid. manufactured to industry standards and finished in black paint.In a lead-acid battery, the cathode is made of lead-dioxide, and the anode is made of metallic lead.All cast equalizers are manufactured from ductile iron.The equalizer is suspended from center hanger (sold separately).Springs attach to ends of equalizer with shackle straps (sold separately).Connects double-eye springs on dual- or triple-axle trailers.Straight equalizer rocker bar connects double-eye springs on tandem- or triple-axle trailers and transfers impact between the axles to ensure that the load is evenly distributed. Straight Rocker Style Equalizer Bar for 1 3/4" Wide Double-Eye Springs, 5 1/2" Hole Centers #32492
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
